NEWS & MEDIA
Shanghai Huaota Biopharmaceutical Co., Ltd. (hereinafter referred to as "Huaota") received FDA approval for the anti-PD-L1/TIGIT bispecific antibody (project code HB0036), which was independently developed by the company, to enter clinical trial. This is also the second bispecific antibody project of Huaota that has been approved by the FDA to enter clinic trial (the first is a PD-L1/VEGF bispecific antibody project). HB0036 is the world's first FDA-approved bispecific antibody targeting two key immune checkpoints -PD-L1 and TIGIT. Dr. Xiangyang Zhu, General Manager of Huaota, expressed his excitement about this project. He stated that HB0036 has excellent CMC properties and excellent anti-tumour effect in preclinical studies. In light of the exciting clinical results of the combination therapy of Atezolizumab and Tiragolumab reported by Roche, he anticipates that HB0036 could bring enhanced benefits to the patients and hopes that clinical study with HB0036 could be launched as soon as possible.
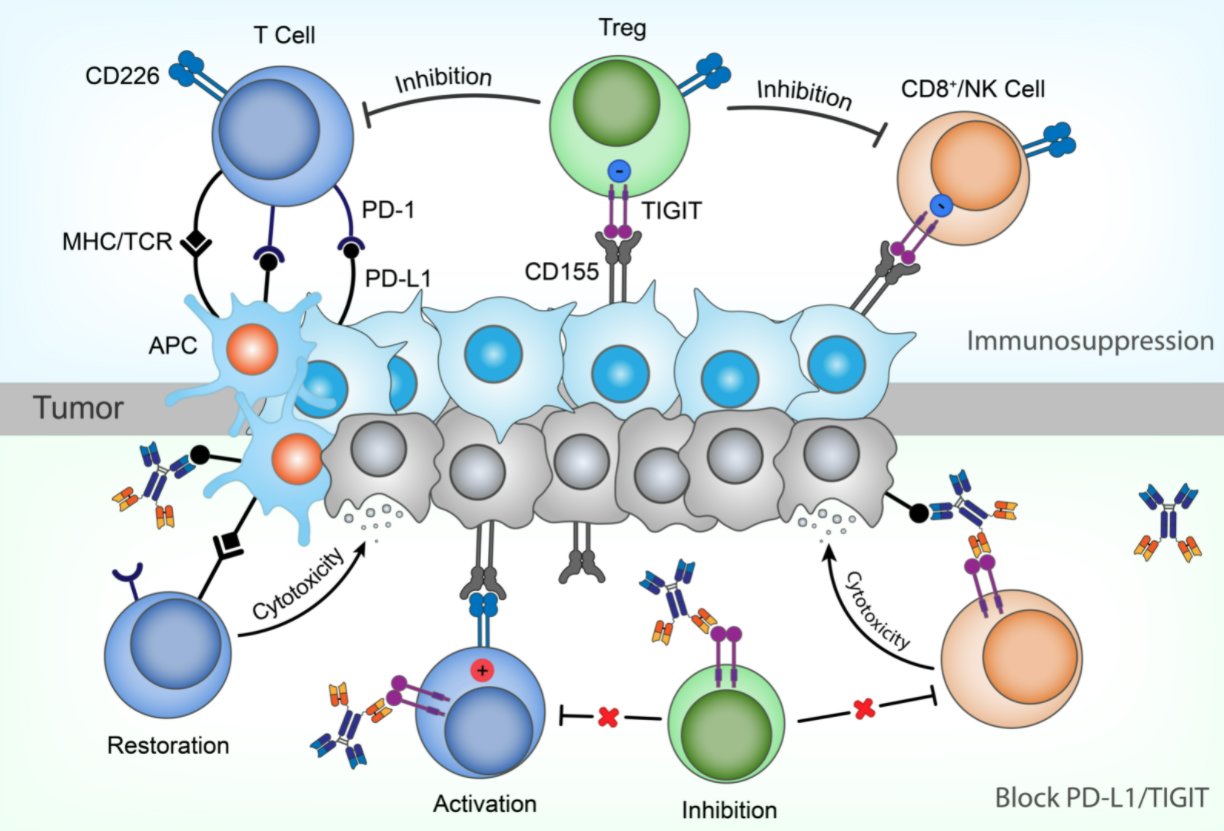
HB0036 is the second bispecific antibody developed by Huaota. It binds to PD-L1 and TIGIT with high specificity and affinity. It has strong anti-tumor effect by blocking both PD-1/PD-L1 and TIGIT/CD155 signaling pathways, ameliorating the immunosuppressive effects mediated by these two signaling pathways and reactivating cytotoxic T lymphocytes and NK cells against tumor cells. In addition, since HB0036 retains the ADCC effect mediated by PD-L1 and TIGIT, it can kill tumor cells and Treg cells, and further enhance the anti-tumor effect.
Preclinical studies have shown that the antitumor effect of HB0036 is better than that of the single drug, and is comparable to the combinational use of two separate antibody drugs. Preclinical pharmacokinetic results in non-human primates showed that HB0036 has linear pharmacokinetic characteristics, long half-life and satisfactory pharmacokinetic properties. Toxicological studies showed that HB0036 is safe and well tolerated, and has great potential to become a new generation of tumor therapy drugs.
There is currently no drug targeting both pathways on the market worldwide. The most advanced competitor drug is the combination of anti-PD-L1 monoclonal antibody Atezolizumab and anti-TIGIT monoclonal antibody Tiragolumab by Roche. In January 2021, the FDA granted Roche's Atezolizumab in combination with Tiragolumab the Breakthrough Therapy Drug (BTD) designation for first-line treatment of NSCLC. Many companies are also conducting research on PD-1/TIGIT bispecific antibody.