NEWS & MEDIA
Shanghai, China, Dec 24 2021 --- Shanghai Huaota Biopharmaceutical Co., Ltd. (Huaota) announced that HB0030 (anti-TIGIT monoclonal antibody) completed the first-in-human administration in China at the First Affiliated Hospital of Bengbu Medical College. This is an open label, single-center, dose-escalation Phase Ia clinical study in order to assess the safety and pharmacokinetics in patients with advanced solid tumors. The main purpose of the study is to comprehensively evaluate the safety, tolerability and antitumor activity of HB0030 in patients with advanced malignant solid tumors.
About TIGIT
TIGIT, T cell immune receptor with Ig and ITIM domains, is an inhibitory receptor expressed on lymphocytes belonging to the Ig superfamily with multi-characteristic structure of immunoregulatory proteins including CD96 and CD226. They interact with Nectin and nectin-like molecules with different affinities.
TIGIT, CD226, CD96 and their ligands CD155 and CD11 form a complex regulatory network. As TIGIT/CD155 deliver the immunosuppressive signal,CD155 interaction with CD226 deliver an immunostimulating signal. The CD155 and CD112 are highly expressed on the surface of many malignant tumors, such as colorectal cancer and melanoma.
About HB0030
HB0030 is a humanized monoclone antibody designed by Huaota Biopharm to bind TIGIT with high-affinity, thereby blocking the binding of TIGIT to its ligand (such as CD155). By binding to TIGIT, highly expressed on the surface of T cell and NK cell, HB0030 blocks its interaction with CD155, relieves immunosuppression and reactivate the tumor-killing effect of T cells and NK cells.
Preclinical studies have shown that HB0030 has higher affinity and strong ADCC/CDC activity. It can relieve the T-cell immunosuppression and enhance the anti-tumor activity.
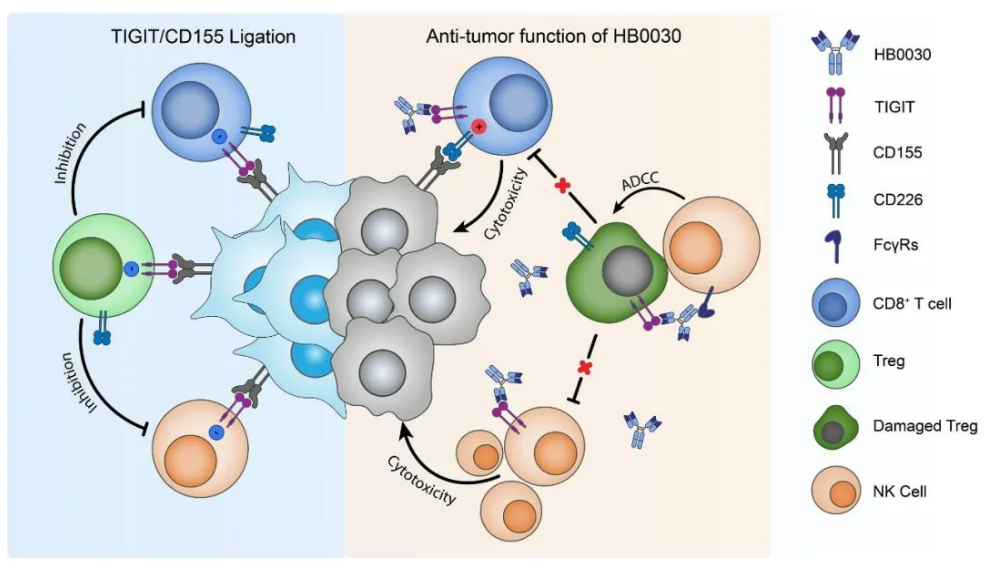
Schematic diagram of the likely action mechanism of HB0030
In the future, HB0030 is planned to use in combination with other antibodies of our company, such as HOT-1030 (CD137 monoclonal antibody) and HB0025 (PD-L1/VEGF bispecific antibody). It is expected that there will be a synergistic anti-tumor effect and achieve extraordinary results.
Market competition
At present, there is no anti-TIGIT antibody in market. The leading candidate of its kind is Tiragolumab from Roche which is in the Phase III clinical trial. In January 2021, Roche announced that Tiragolumab has been granted Breakthrough Therapy Designation (BTD) by the US Food and Drug Administration (FDA), in combination with Atezolizumab for the first-line treatment of people with NSCLC whose tumours have high PD-L1 expression with no EGFR or ALK genomic tumour aberrations. Vibostolimab, anti-TIGIT monoclonal antibody developed by Merck, is also under Phase III clinical study with the combination use of Pembrolizumab (PD-1 monoclonal antibody) to treat NSCLC. As an emerging immune checkpoint in cancer immunotherapy, TIGIT has great potential becoming a gamechanger and benefits those patients who shows resistance or poor response to current immunotherapy. The future clinical application of anti-TIGIT antibodies is worth exploring.